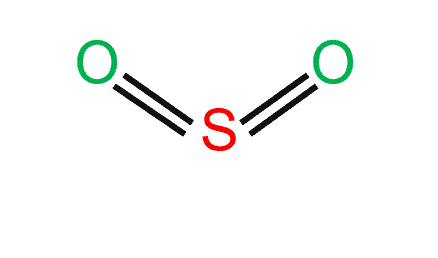
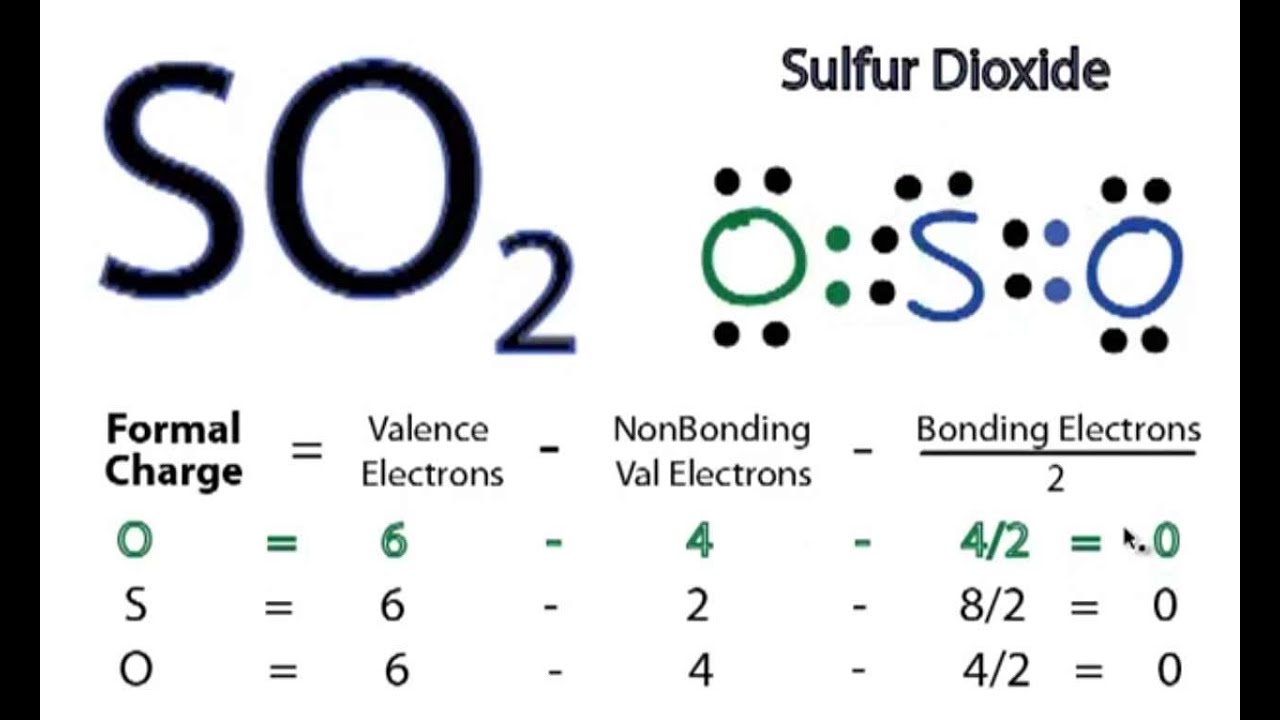
SO2 Lewis Structure||Sulfur dioxide Lewis Structure ||Lewis Structure for SO2 | Chemistry worksheets, Chemistry notes, Lewis

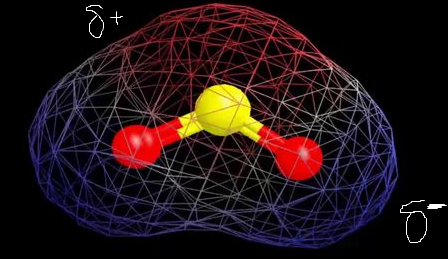
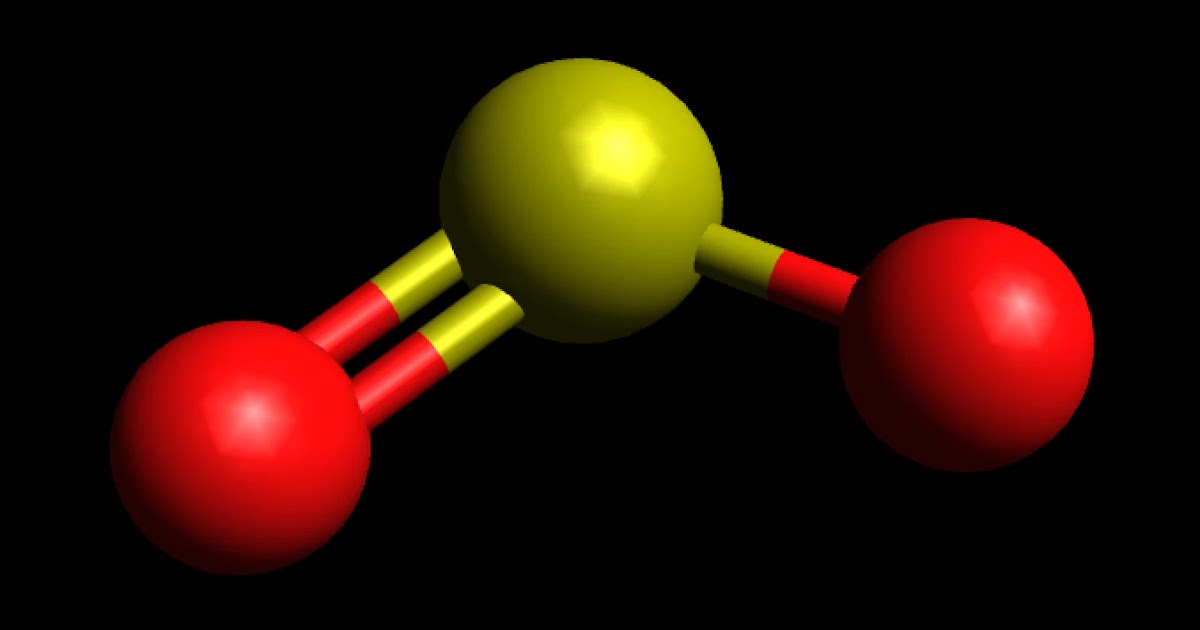
Why is carbon dioxide non-polar even though the C=O bonds are polar bonds but SO_2 is a polar compound? | Homework.Study.com

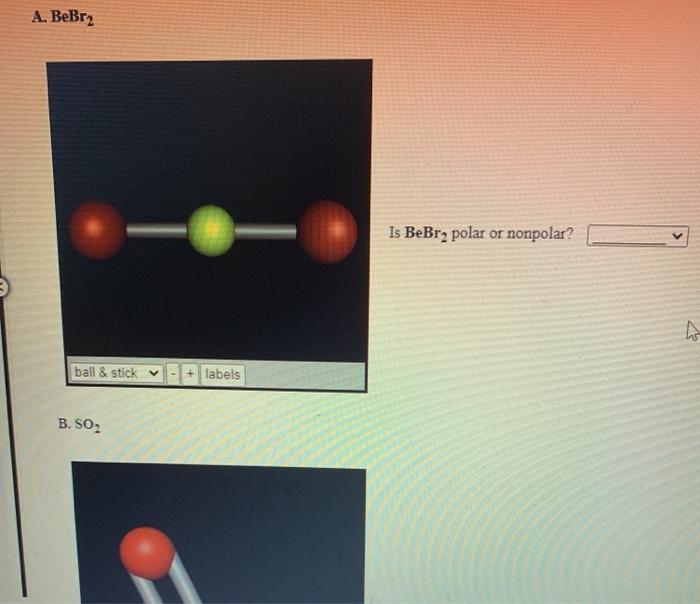
Determine the following for SO2. a. Electron geometry b. Molecular geometry (shape) c. Is the molecule symmetrical or asymmetrical? d. Is the molecule polar or nonpolar? | Homework.Study.com