A) Structure of NaF (Na-large size (red) sphere, F-small size (blue)... | Download Scientific Diagram
![Draw the electron dot diagram of the following compounds. 1. NaF 2. MgF2 [Hint: Atomic No. Na = 11, F = 9, Mg = 12 ] Draw the electron dot diagram of the following compounds. 1. NaF 2. MgF2 [Hint: Atomic No. Na = 11, F = 9, Mg = 12 ]](https://haygot.s3.amazonaws.com/questions/1758756_1836814_ans_d4bea0106ad5432db73cf0c7388b667d.png)
Draw the electron dot diagram of the following compounds. 1. NaF 2. MgF2 [Hint: Atomic No. Na = 11, F = 9, Mg = 12 ]

⚗️NaF + A. Br2 NaBr + F2 Na= F= Na= F= 1. Use equation A to choose the correct coefficients from the - Brainly.com

SOLVED:Oxidation Number Exercise pag? 51 Rule 2 Fluorine has an oxidation number of Exercises Give the oxidation number for the following atoms: NaF Na CIF; Cl = SF4 PF, SF& PFs PF;*
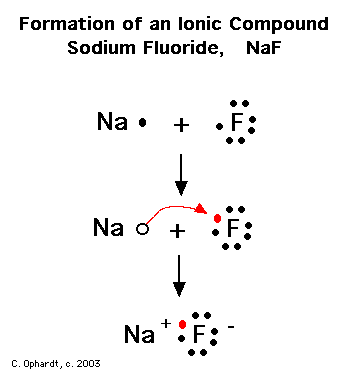
The strongest interactions in the compound sodium fluoride, NaF, are an example of what type of bond? | Socratic
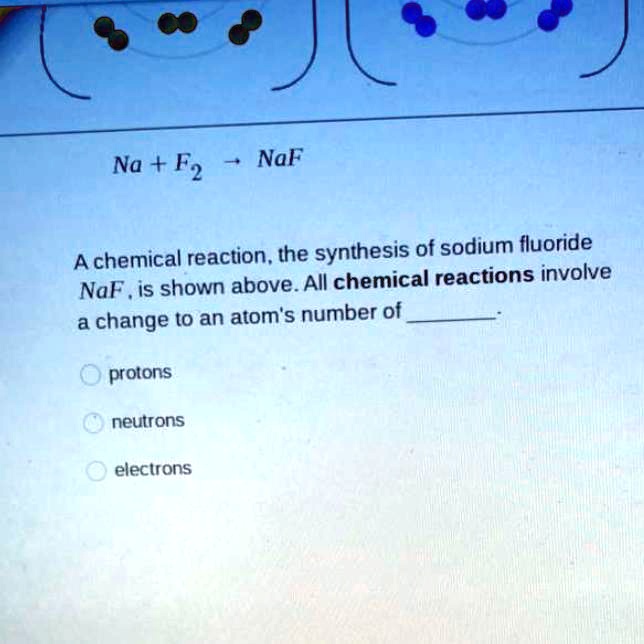
SOLVED:"na+F2-> naF a chemical reaction. the synthesis of sodium fluoride noF is shown above all chemical reactions involve a change to an atoms number of protons neutrons electrons NaF Na + F2